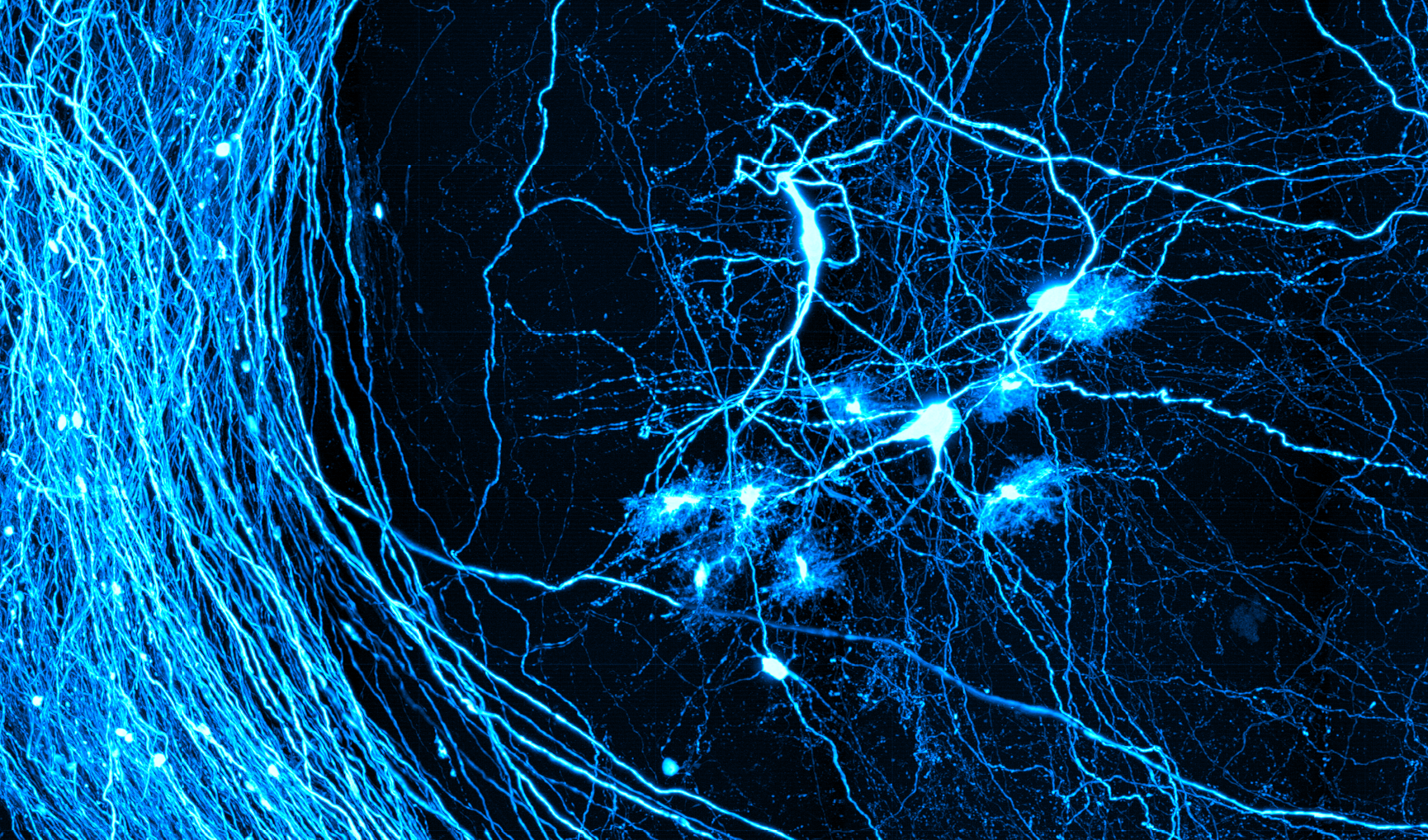
Gabel Lab: Neurophotonics in Simple Systems
Put simply, the Gabel Lab employs light to study neuroscience in worms. C. elegans, a microscopic nematode, or roundworm, that is completely transparent, exhibits characteristics that make it ideal for photonic interrogation of the nervous system. The microscopic nematode possesses the simplest of nervous systems and is genetically tractable with a vast library of well-defined mutants that allow the expression of genetically encoded fluorophores in select neuron types. The C. elegans nervous system consists of 302 stereotyped neurons whose anatomy and connectivity is completely mapped. To this system the Gabel lab brings expertise in high-resolution laser ablation and fluorescence microscopy to study neuronal damage, regeneration and function. Dr. Gabel’s lab is also very active in the Neurophotonics Community. Each year, the lab hosts our newest cohort of the National Science Foundation Research Traineeship Program, Understanding the Brain: Neurophotonics (NSF NRT UtB: Neurophotonics). The Gabel lab runs one of the intensive half-day workshops in boot camp and each year, trainees in his lab assist in running hands on sessions with C. elegans.

With a background in physics and biophysics, Dr. Gabel was attracted to the powerful simplicity of C. elegans and similar model systems. As a graduate student in Physics at Harvard University, he worked with Dr. Howard Berg, using biophysical techniques to study the functional physiology of the bacterial flagellar motor. As a post-doc, Dr. Gabel worked with Prof. Aravinthan D.T. Samuel from the Department of Physics & Center for Brain Science at Harvard. Here, he was first introduced to C. elegans, as the simplest most tractable model organism in neuroscience. During his postdoctoral work, he helped develop the high-resolution laser ablation techniques currently used in his lab, as well as expertise in neuronal imaging and quantitative behavioral analysis.
Laser surgery and regeneration of single neurons
Employing a pulsed infrared laser, the Gabel lab performs subcellular ablations within bulk biological tissue. The super-high intensity of the tightly focused femto-second pulsed laser beam results in multi-photon absorption and vaporization of the tissue within the focal volume. At the same time, the low total energy of each light pulse minimizes collateral damage to the surrounding tissue. The minimal linear absorption of the infrared light enables deep beam penetration. Applied to C. elegans this technique enables selective severing of individual nerve fibers within intact adult animals.

Neurological damage presents a major medical challenge, as severed neurons within the mammalian central nervous system typically do not regenerate resulting in permanent and debilitating paralysis. With C. elegans, nerve regeneration from neuronal damage induced by laser surgery can be studied in a simple highly tractable and reproducible system. The animal’s simple, completely mapped, nervous system allows identification, targeting and severing of a particular neuron at a specific location. Subsequent time-lapse imaging quantifies the degree of regeneration that can be compared across genetic backgrounds, pharmacological treatments and experimental conditions. Armed with these tools the Gabel lab has performed a series of studies decoding the cell intrinsic mechanisms of neuronal damage, recovery and regeneration. Their studies have defined the role of apoptotic caspases (nominally known for mediating programed cell death during development) in regeneration. As a post-doc in the Gabel lab, Dr. Samuel Chung, currently an Assistant Professor of Bioengineering at Northeastern University, led a study defining the effects of neuronal activity on regeneration. More recently, in a project initiated by Dr. Dan Taub, a former graduate student in the lab, the group has focused on the fundamental importance of cellular metabolism in dictating a neurons regenerative capacity. Notably, they have discovered post-translational protein modifications that can alter cellular metabolism to greatly enhance neuronal regeneration.
Neuronal imaging from single cell physiology to whole brain function
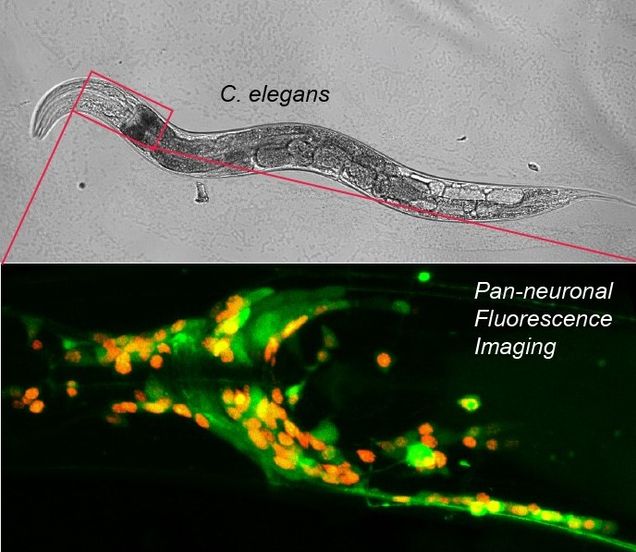 The Gabel lab has adapted the use of genetically encoded fluorophores and in vivo imaging of calcium dynamics to measure subcellular calcium dynamics within a damaged and regenerating neuron in C. elegans. By laser severing and imaging a neuron expressing calcium sensitive G-CaMP, they have identified sustained sub-cellular calcium signals and the modulating cellular components. These cellular components are critical to efficient regeneration. Moreover, by employing optogenetic light activated channels, the Gabel lab is able to manipulate these signals, enhancing the neurons regenerative capacity. Using modern neurophotonic techniques, the Gabel lab can identify and discretely damage an individual neuron, measure its physiological response, and modulate that response to improve neuronal recovery.
The Gabel lab has adapted the use of genetically encoded fluorophores and in vivo imaging of calcium dynamics to measure subcellular calcium dynamics within a damaged and regenerating neuron in C. elegans. By laser severing and imaging a neuron expressing calcium sensitive G-CaMP, they have identified sustained sub-cellular calcium signals and the modulating cellular components. These cellular components are critical to efficient regeneration. Moreover, by employing optogenetic light activated channels, the Gabel lab is able to manipulate these signals, enhancing the neurons regenerative capacity. Using modern neurophotonic techniques, the Gabel lab can identify and discretely damage an individual neuron, measure its physiological response, and modulate that response to improve neuronal recovery.
The Gabel lab has recently developed techniques to measure neuronal activity in C. elegans within medically relevant context such as physical damage, age related decline and exposure to anesthetics. Neuron activity is measured optically with calcium indicators reporting the elevation of cellular calcium resulting from neuronal depolarization. In collaboration with Professor Christopher Connor in the Department of Anesthesiology at Brigham and Women’s Hospital and Harvard University, the lab recently acquired a diSPIM light sheet microscope capable of fast volumetric imaging of the entire head of C. elegans. The small size of its nervous system and cell specific expression of calcium indicators allows imaging of individual neurons, specific well-defined circuits controlling behavior or the activity across the majority of the nervous system with single cell resolution.
Gregory Wirak, a fourth year Ph.D. Candidate in the Graduate Program for Neuroscience and an NSF NRT UtB: Neurophotonics trainee, is leading studies in the Gabel lab on the chronic effects of volatile anesthetics on neurodevelopment and how aging affects neural circuits. Gregory graduated from the University of Connecticut in 2013, with a B.S. in Physiology & Neurobiology and Molecular & Cellular Biology. He then spent three years engaged in postgraduate research in the laboratory of Dr. Sreeganga Chandra at Yale University. There he used biochemical techniques and human stem cell-derived and primary murine neuronal cultures to study the neuronal ceroid lipofuscinoses, a group of inherited neurodegenerative disorders. Upon joining Dr. Gabel’s lab as a graduate student, he discovered the many advantages granted by the stereotyped C. elegans nervous system and its tractability for functional in vivo research. Greg has recently discovered that exposure to volatile anesthetics during development alters the neuronal dynamics of key interneurons in the adult animal. These findings open a new window into the potential health risks of anesthetics in the very young. In addition to his involvement in the NSF NRT Neurophotonics community as a trainee, Greg is also engaged in the NIGMS Biomolecular Pharmacology Training Program.

Mehraj Awal, one of Dr. Gabel’s graduate students and also an NSF NRT UtB: Neurophotonics Trainee, received his Bachelor of Science in Biology and Biochemistry from Brandeis University in 2014. Shortly thereafter, he entered the Boston University Program in Biomedical Sciences (PiBMS). Upon joining the Gabel lab, Mehraj has played a critical role in initiating the studies to investigate the acute effects of volatile anesthetics on neural circuits in C. elegans. The group’s initial publication on this project demonstrates that neurons within a highly ordered sub-circuit of C. elegans that control behavior become disorganized and hyperactive with the onset of anesthesia. Moving forward, Mehraj seeks to further this work and apply it to a greater number of neurons using light sheet microscopy, in hopes of bridging the gap between global brain dynamics and small neuronal networks. This will generate a novel understanding of volatile anesthetics on a systems level and how this correlates with measurements made on human patients. Ultimately, functional neuronal imaging in C. elegans presents an opportunity to understand how volatile anesthetics disrupt neuronal function in a controlled and reversible manner.
More information about Dr. Gabel’s laboratory and research areas can be found at http://www.bumc.bu.edu/phys-biophys/people/faculty/gabel/.