Diagnostics
Alan and Sandra Gerry Amyloid Reference Laboratory
The Gerry Amyloid Reference Laboratory performs specialized diagnostic testing for systemic amyloidosis. Our goal is to provide patients and physicians with accurate and timely diagnosis of the amyloid disease. While we only provide genetic testing for our own patients and their families, please contact the BMC Pathology department if you are interested in sending samples for Congo red staining or immunohistochemistry. BMC Pathology can be contacted by calling 617-638-6994 and to discuss the cost of testing please contact Gail Whitney at 617-414-4291. Please be aware that the cost of testing may be covered by insurance or a referring hospital, or may have to be paid for individually. We are not able to bill insurance outside of Massachusetts.
Fat Aspirates, Biopsies, and Congo red Staining for Diagnosis of Amyloidosis
Material from a fat aspirate or organ biopsy can be stained with the Congo red dye and examined by polarized light microscopy, under which amyloid fibrils have a typical green birefringence. There are charges for these services.
Histopathology Techniques for Amyloid Typing
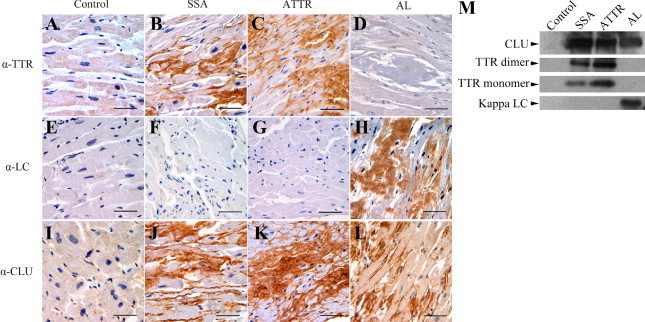
We can perform immunohistochemistry for immunoglobulin light chains (kappa and lambda), transthyretin (TTR), serum amyloid A (AA), apolipoproteins AI and AII, lysozyme, and fibrinogen. In some cases, immunoelectron microscopy using immunogold antibodies may be employed. Biopsies obtained for possible immunoelectron microscopy should be fixed in 4% paraformaldehyde rather than standard formalin. Charges for this service depend upon how many antibodies are needed for typing .
Molecular and Genetic Testing
For patients suspected of having an inherited or familial form of systemic amyloidosis, the Gerry Laboratory performs molecular and genetic testing techniques. These analyses are generally carried out on blood samples.
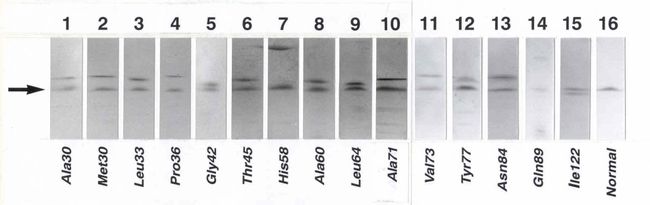
- The testing for in-patients includes isoelectric focusing screening for TTR variantsand PCR-based gene sequencing for the familial types of amyloidosis (TTR, fibrinogen, lysozyme, apolipoproteins AI and AII, and gelsolin). Charges for molecular and genetic testing depend upon what is required for each case.
- The genetic testing for out-patients is offered through The testing can be ordered by primary care provider or other specialist using Upon the completion of genetic testing, Invitae offers genetic counseling to interpret the results with patient. Amyloidosis Center staff would be available to discuss next steps of patient care should the genetic test result be positive.
- Research-related genetic testing includes PCR-based sequencing for genes coding for the following amyloidogenic proteins: apolipoproteins AI, AII, CII, and CIII, beta 2 microglobulin, fibrinogen, gelsolin, leukocyte cell derived chemotaxin 2, lysozyme, and TTR.
Based upon recent federal legislation, patients who have hereditary predisposition to diseases like amyloidosis cannot be discriminated against with respect to employment or health insurance. For more information, search the web for information on GINA, the “Genetic Information Nondiscrimination Act”.
General Information
The Amyloid Reference Laboratory is accredited by the College of American Pathologists under the aegis of the Pathology and Laboratory Medicine Department of Boston Medical Center. Tatiana Prokaeva, MD, PhD directs the Gerry Amyloid Reference Laboratory and Christopher D. Andry, PhD, is Chief and Chair, Department of Pathology and Laboratory Medicine.
Please contact Amyloidosis Center administrative office at 617-358-4750 or amyloid@bmc.org if you have questions.