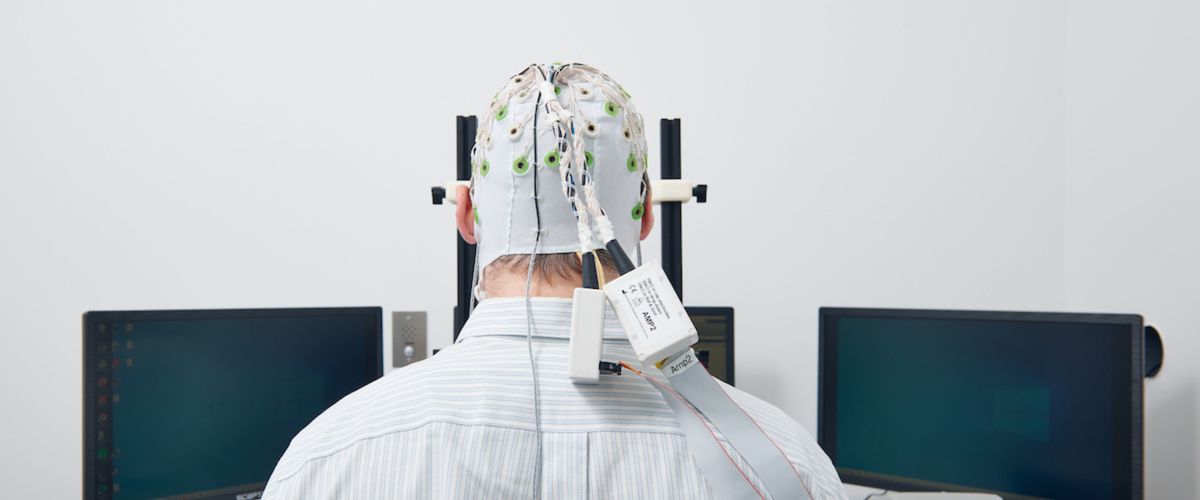
Global Reliance Agreements Established Between CRC, BMC, and BUMC
Boston University’s IRBs have put in place global reliance agreements to streamline multisite collaboration across the Charles River Campus (CRC), Boston Medical Center (BMC) and Boston University Medical Campus (BUMC). These agreements: Automatically apply to qualifying minimal‑risk collaborative research Simplify and standardize the reliance process for faculty and staff working across the campuses Take effect […]