Work It Out
Can exercise help delay or prevent Alzheimer’s disease?
Alzheimer’s disease is an epidemic. It attacks the brain’s nerve cells, causing memory loss, behavioral changes, confusion, and deterioration of language skills. It affects more than 5 million Americans 65 and older, and that is expected to increase to 13.8 million by 2050 unless science finds a treatment. Alzheimer’s and other forms of dementia are projected to cost the nation $236 billion this year and the figure could reach $1 trillion by 2050, according to the Alzheimer’s Association.
At Boston University, dozens of researchers are looking for tests that could lead to early diagnosis and interventions to prevent or delay the disease, running clinical trials that may result in treatments and an eventual cure, working to understand genetic risk factors, and studying Alzheimer’s impact on caregivers.
The Boston University Alzheimer’s Disease Center, established in 1996, is one of 31 such centers nationwide funded by the National Institutes of Health and dedicated to conducting research into the disease, enhancing clinical care, and providing education.
In this special report, BU Today examines the work of five BU researchers.
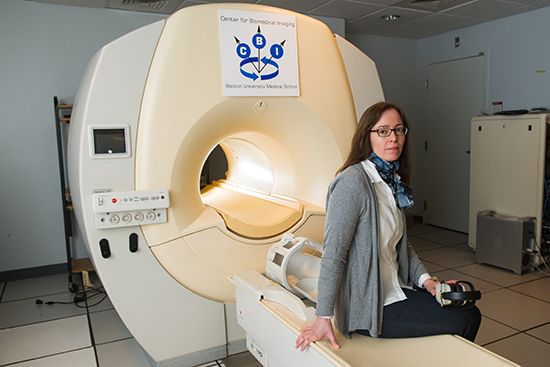
When Karin Schon enrolled in a 12-week fitness training program during her doctoral studies in cognitive neuroscience at BU, she expected it to improve her health. But she had no idea that it would also shape her research.
Before she took a single step, her trainers gave her a fitness test that measured body fat, weight, and cardiovascular fitness. That first test provided a baseline; another test three months later would determine if the program had made a difference. “I liked it from a scientific perspective,” says Schon (GRS’05), a School of Medicine assistant professor of anatomy and neurobiology, and a BU Alzheimer’s Disease Center faculty member, who studies the effects of physical activity on the aging brain. “It felt like I was participating in a study.”
The program worked. She lost about 20 pounds, her cardiovascular fitness improved, and she was able to hold plank (the upper position of a push-up and a test of core strength and endurance) for nearly seven minutes.
“This program had such an impact on me physically,” says Schon, who later became an accomplished half-marathon runner. “I was also curious. I thought that it must have an effect on the brain if it has an effect on the body.”
Schon’s fitness program coincided with her graduate work and prompted a new research question: What effect does exercise have on the brain, particularly on memory?
Studies in animals showed that increased levels of exercise, such as running on a wheel, improved the function of the hippocampus. But Schon found almost no studies of the effects of exercise on the human brain. “I started to connect the dots,” she says. “And I found a gap.”
She decided to study the effects of exercise on memory, and in 2010 won a Pathway to Independence Award from the National Institute on Aging, which provided five years of funding to pursue her idea. “Exercise should be great for healthy aging,” she says. “If exercise improves memory in healthy adults as it does in animals, maybe it can be a way to ameliorate cognitive decline.”
In the United States today, cognitive decline is a serious concern. One in three senior citizens dies with Alzheimer’s disease or another form of dementia and the number of cases is rising, according to the Alzheimer’s Association. The costs of caring for people with Alzheimer’s is expected to be approximately $1 trillion by 2050. These numbers are staggering, and given that the US health care system is already straining to meet health care demands, rather frightening.
Schon’s idea, however, could change the equation. What if something as simple as a brisk daily walk delayed the onset of Alzheimer’s disease? Since she started looking into this in 2008, the number of studies suggesting a connection between exercise and the human brain has increased dramatically. “This is about lifestyle changes,” says Rhoda Au (Questrom’95), a MED professor of neuropsychology and a School of Public Health professor of epidemiology, who is one of Schon’s mentors and collaborators. “If you can make healthy lifestyle changes early on, maybe you can prevent disease later on. There’s nothing that’s going to reduce health care costs faster than removing the need for it.”
Schon studied psychology as an undergraduate at the University of Hamburg in Germany, and afterwards the aging brain during a stint at the National Institutes of Health. As a graduate student at BU, she joined the lab of neuroscientist Chantal Stern, a College of Arts & Sciences professor of psychological and brain sciences, and learned to use functional magnetic resonance imaging (fMRI) to study the hippocampus, a seat of memory in the brain.
To measure memory capabilities, Schon showed study participants pictures in a series. She presented one image, then another, then a test picture, with delays between each presentation. The participants determined if they had seen the test picture before, as in the game Concentration.
She’s found that the fMRI patterns during the delays predict whether or not someone remembers the picture 15 minutes later. “It makes sense,” she says. “If I just saw this picture, maybe I’m thinking about it for awhile.”
She used this method to study the relationship between fitness and memory. She recruited a group of students from BU to take a fitness test and then look at pictures in the fMRI scanner. She discovered an indirect connection between fitness and memory performance. A growth hormone called BDNF appears to connect the two. “There’s greater memory accuracy with greater levels of BDNF in more fit individuals,” explains Rachel Nauer (GRS’15,’20), a graduate student, and a marathon runner, in Schon’s Brain Plasticity and Neuroimaging Laboratory.
BDNF is also known to play a role in the growth of new neurons in the brain. This discovery provided Schon with the first of several connections between her research and Alzheimer’s disease.
Making a connection to Alzheimer’s disease in any research program is difficult. The disease, even with all that’s known about it, is still mysterious. It occurs when a protein called amyloid forms plaques in the brain. Tangles, caused by tau, a different protein, follow, as does neurodegeneration, the loss of brain cells.
It isn’t clear yet what causes plaques to form in the first place, or if those plaques directly cause tangles to appear. Also, plaques may begin forming in the brain 20 years before a person begins to misplace car keys or forget names. This long lead time makes it extremely difficult to study the progression of the disease and especially difficult to show that an intervention slows progression.
Enter Jake Gyllenhaal
So instead of focusing on Alzheimer’s disease directly, Schon is focusing on links to it in the brain. One link is BDNF, which helps the brain grow new neurons. In the hippocampus, that neurogenesis occurs in a subregion called the dentate gyrus. The dentate gyrus is of particular interest to Schon because exercise enlarges it in animal studies.
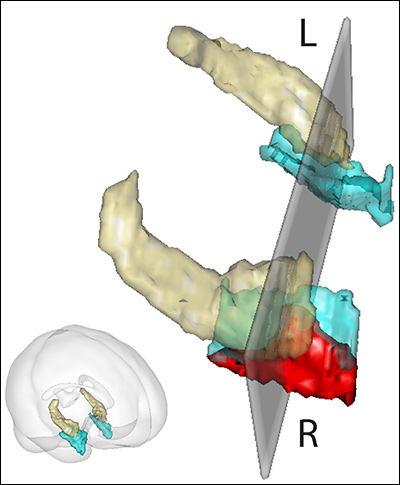
A second link to Alzheimer’s disease is right nearby, in the entorhinal cortex, which is connected to the dentate gyrus. Schon’s team recently found that the entorhinal cortex is bigger in people with higher fitness levels. Animal studies suggest that these two regions of the brain—the dentate gyrus and the entorhinal cortex—malfunction first in Alzheimer’s disease. “We started thinking it would be really interesting to look at these brain regions in the context of an exercise intervention in humans,” Schon says.
She plans to study the effects of exercise on the entorhinal cortex, pending NIH funding. She is also studying an exercise intervention focusing on the dentate gyrus. To do this, she and Nauer needed to develop a method to specifically stimulate the dentate gyrus. What they needed was an extremely sensitive test to detect changes in this tiny brain region, and potentially, to connect them to subtle behavior changes that other cognitive tests miss.
The new test Schon’s group developed builds on Schon’s earlier fMRI methods for stimulating the hippocampus. The team presents images in a series, but they use special images that trigger the brain to perform a process called pattern separation. Pattern separation deciphers subtle differences in images and—critically—it occurs in the dentate gyrus. So they believe the method triggers activity in a seat of neurogenesis in the brain, and a place of earliest vulnerability in Alzheimer’s.
Nauer illustrates the types of images they show using familiar faces, such as actors Jake Gyllenhaal, Joseph Gordon Levitt, and John Travolta, although in the actual experiments, the images are of unfamiliar scenes. In her example, she shows faces that have been morphed using computer software so that they have specific degrees of similarity. One face might be 90 percent Gyllenhaal and 10 percent Levitt, for example, and another might be 90 percent Levitt and 10 percent Gyllenhaal. As the degree of similarity increases, the images become harder to decipher.
They are still recruiting young people age 18 to 35 and older people age 55 to 85 who are currently sedentary for the study. Participants enroll in a fitness program similar to the one Schon took years ago. Nauer and Matt Dunne, a research study technician in the Schon lab, give each participant aerobic and muscle function tests and a series of memory tests, including the pattern separation test in the fMRI. They also sample BDNF levels.

The participants are then given an individualized exercise prescription—either an aerobic fitness routine or a resistance training routine—to follow 3 times a week for 12 weeks. Since animal studies indicate that cardiovascular fitness is the key to improved memory, the researchers consider the resistance training group to be a control.
One of the things that makes Schon’s research so powerful is its interdisciplinary nature. For instance, she’s brought in an exercise physiologist from Brigham and Women’s Hospital, Thomas Storer, to evaluate the exercise tests and prescribe the fitness routines. “Both training programs start out at a level that is doable for the individual and then progress over time with more added challenge,” says Storer. “We can get a good training effect even with just three days of training per week.”
Storer’s input adds heft to the program. “Studies like these tend to be done either by neuroscientists who don’t really have the exercise background or people with the exercise background who don’t really know the neuroscience,” says Dunne, a former college soccer player. “We’re combining the two so we can have a rigorous study from both perspectives.”
For Nauer, this research isn’t so much about whether exercise will make a difference, but what kind of exercise makes the most difference and how much is needed. “My focus moving forward is doing research that informs doctors,” she says, “so they may someday prescribe an exercise program rather a drug.”
Putting a dent in the problem
Schon is also working with Au on another exercise intervention, which uses digital cognitive assessments to detect subtle memory changes. These assessments not only record answers to memory questions, but also track the thinking that goes on behind these tasks. The intent of the research, says Au, is to identify changes in behavior that are more subtle than the symptoms a doctor would look for.
This distinction between disease treatment and disease prevention is a critical one. Exercise may help individuals with cognitive impairments slow the disease, but Au thinks Schon’s work could have much wider implications. The way she sees it, if exercise prevents cognitive decline, it could be a way to shrink the looming specter of Alzheimer’s disease.
Since 1990, Au has been an investigator for the Framingham Heart Study, the nation’s longest running epidemiological study, begun in 1948, supported by the National Heart, Lung, and Blood Institute, and run by BU since 1971. According to a study report, delaying Alzheimer’s disease by five years reduces a person’s risk of getting the disease by 50 percent. “Essentially, the disease is competing with mortality,” she explains.
If fitness delays Alzheimer’s disease enough to reduce the risk by any percentage, that puts a dent in the problem. With the number of Americans with Alzheimer’s possibly reaching 7.1 million in the coming decade, any dent is welcome.
While Au sees the big picture potential, Schon’s eyes are necessarily on the science. She’s busy writing grant applications, mentoring students, engaging new collaborators, and reading and writing papers. She even took time to take an undergraduate exercise physiology class during her postdoc to round out her skill set.
She’s also getting back into running. She had knee surgery seven months ago and is just about fully recovered. “I don’t like the status quo right now, not being able to run,” she says. “My last half marathon was two years ago. I’d love to be able to do that again.”
November has been designated as National Alzheimer’s Disease Awareness Month.
Elizabeth Dougherty can be reached at beth@writtenbyelizabethdougherty.com.
Read the other stories in our “Unraveling Alzheimer’s Disease” series here.
This Series
Also in
Unraveling Alzheimer’s Disease
-
April 29, 2016
Caring for the Caregivers
-
April 28, 2016
The Gene Hunter
-
April 25, 2016
Diagnosis: Alzheimer’s








Comments & Discussion
Boston University moderates comments to facilitate an informed, substantive, civil conversation. Abusive, profane, self-promotional, misleading, incoherent or off-topic comments will be rejected. Moderators are staffed during regular business hours (EST) and can only accept comments written in English. Statistics or facts must include a citation or a link to the citation.