Current research
Of the millions of lives lost each year to cancer, 90% are due to tumor aggressiveness and distant spread. There is an urgent need to develop novel treatment alternatives that can efficiently target and destroy these aggressive cancer cells. MYC, aptly referred to by Gerard Evan as “the oncogene from hell”, is aberrantly activated in nearly all human cancers, including leukemias, lymphomas, and carcinomas. Cancers with aberrant MYC/MYCN activity are often aggressive, rapidly spreading to distant tissues. These facts underscore the need to 1) improve our understanding of the regulatory molecules controlling MYC/MYCN-mediated tumor initiation and progression, 2) discover the vulnerabilities of MYC/MYCN-driven cancers, and 3) develop novel and effective therapeutics by exploiting the vulnerabilities of MYC/MYCN-driven cancers.
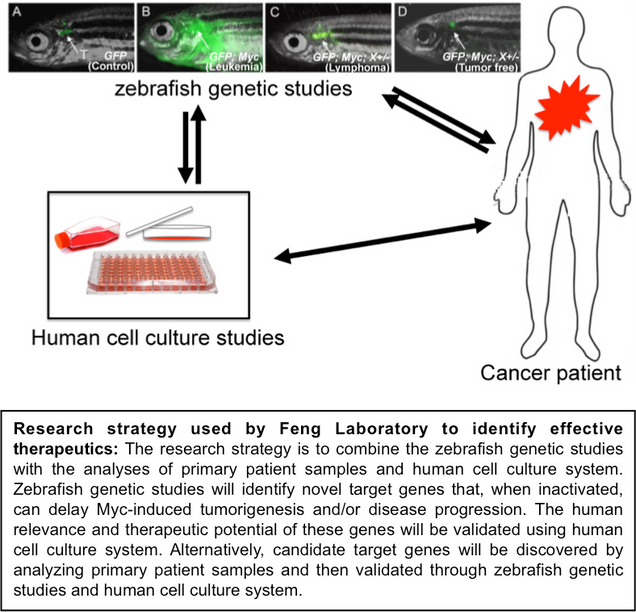
The research in the Feng Laboratory combines the analyses of human cancer cell lines and patient samples with the animal models (zebrafish and mice) to:
1) discover novel genes and pathways essential for MYC/MYCN-mediated tumor initiation and progression;
2) screen for and develop chemical compounds that can suppress MYC/MYCN-driven tumor development;
3) evaluate their potential as therapeutic targets or leads.
We included the zebrafish model of cancer as an innovative experimental system in our research to accelerate novel gene and compound discovery. The zebrafish offers many unique advantages as a cancer model: easy monitoring of tumor development in vivo, due to its transparency and the ability to differentially fluorochrome-label tumor cells and vasculature; a high degree of genetic similarities with humans; advanced technologies for functional studies of candidate genes identified through ongoing analysis of human cancer genomic databases; and the feasibility of conducting genetic and chemical screens to dissect the molecular pathways of tumorigenesis and identify promising lead anti-neoplastic compounds.
A major undertaking in our laboratory is to dissect molecular pathways that regulate tumor aggressiveness and progression. Improved understanding of genes and pathways governing tumor progression is the key to future therapeutic development, to block the spread of tumor cells from their primary site and inhibit disease aggressiveness. By interrogating human cancer genomic information with the imaging and genetic capacities of the zebrafish system, we focus on identifying novel genes that promote the spread of MYC/MYCN-driven tumor cells. Using this combined approach, we have successfully identified multiple promising genes whose expression is elevated in disseminating cancers, compared to their localized counterparts. We predict that genes identified through our studies will represent promising targets for therapeutic intervention to block MYC/MYCN-driven tumor progression.
The second project in our laboratory is the exploitation of the forward genetic capacity of the zebrafish system in order to identify genes that, when inactivated, can inhibit or delay tumor induction and/or progression. In conducting a forward genetic suppressor screen, we have identified a gene critical for energy production and macromolecule synthesis, dihydrolipoamide succinyltransferase (DLST), and a gene that sustains the survival of MYC-driven cancers, ubiquitin fusion protein 1 (UFD1) by mitigating their ER stress. We collaborate with chemists to develop therapeutic strategies and compounds to target these cancer-dependent pathways.
The third project in our laboratory is to take advantage of the imaging advantages of the zebrafish and its conservation in hematopoietic development to study immune cell development and tumor immunology. We have demonstrated that the tumor microenvironment (TME) in zebrafish resembles their human counterparts, allowing us to leverage genetic and chemical biology approaches to identify novel immunotherapeutic targets.
In the short-term, the Feng Laboratory will focus on identifying and characterizing genes and pathways important for the initiation, maintenance, and/or progression of MYC-induced T-cell lymphoma, MYCN-driven neuroblastoma, and breast cancer, and testing the potentials of inhibiting these genes or related pathway regulators to treat these diseases. In the long-term, we will extend these studies to include other MYC/MYCN-dependent cancers and collaborate with industry and clinicians to develop novel cancer drugs.