Imaging technique solves long standing mystery in fighting fungal infections

By Alex LaSalvia
Fungal infections are estimated to be currently affecting nearly a billion people around the world, with severity ranging from asymptomatic to life threatening. Battling these infections is one of the most important public health challenges today, and the medication amphotericin B (AmB) is a powerful first line of defense against dangerous fungi. Though this treatment has been around for more than 50 years, it’s been unknown exactly how AmB attacks fungus cells until a recent breakthrough from Boston University Professor Ji-Xin Cheng (ECE, BME, MSE) and his team, published in Science Advances, found the answer scientists have been searching for, which could lead to the development of new treatments for fungal infections.
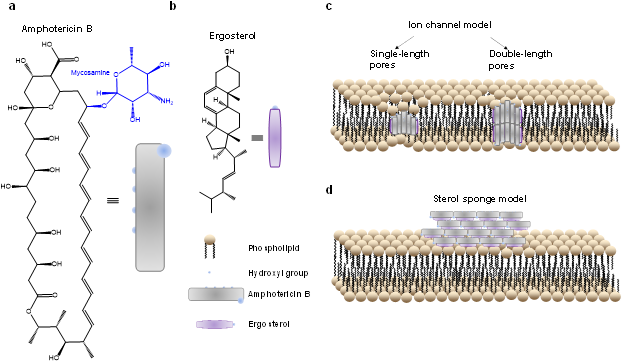 AmB is effective at fighting fungal infections because it infiltrates fungus cells, and its high toxicity kills them. But until now, despite the existence of two theoretical models, the drug’s method of infiltrating the cells’ membranes was unknown. The older model proposed that the AmB molecules orient vertically and create a transmembrane “ion-channel” tunnel. A more recently proposed “sterol sponge” model suggests that the AmB molecules orient laterally to the cell surface and get absorbed into the membrane. To solve this mystery, Cheng and his team developed an imaging technique called “polarization-sensitive stimulated Raman scattering” to see how the drug interacts with fungus cells with minimal interference. In doing so, they found that the AmB molecules were highly ordered in a primarily vertical orientation relative to the membrane, supporting the ion-channel model.
AmB is effective at fighting fungal infections because it infiltrates fungus cells, and its high toxicity kills them. But until now, despite the existence of two theoretical models, the drug’s method of infiltrating the cells’ membranes was unknown. The older model proposed that the AmB molecules orient vertically and create a transmembrane “ion-channel” tunnel. A more recently proposed “sterol sponge” model suggests that the AmB molecules orient laterally to the cell surface and get absorbed into the membrane. To solve this mystery, Cheng and his team developed an imaging technique called “polarization-sensitive stimulated Raman scattering” to see how the drug interacts with fungus cells with minimal interference. In doing so, they found that the AmB molecules were highly ordered in a primarily vertical orientation relative to the membrane, supporting the ion-channel model.
“We can see the drug for the first time,” Cheng said.
Now that scientists know how AmB infiltrates fungal cells, they can develop new treatments that use the same infiltration method. This is important because AmB is a highly toxic drug, and although it has a higher binding affinity for ergosterol than cholesterol, making it highly-effective against fungal cells, it still produces negative side effects such as nephrotoxicity and electrolyte disturbances.
Cheng said his research is ongoing and they will continue to work to fully understand fungal infections and how to treat them.
Cheng is a professor in the Departments of Electrical and Computer Engineering as well as Biomedical Engineering in the College of Engineering at Boston University. For his pioneering contributions to innovation, discovery, and clinical translation of chemical imaging technologies, Cheng received the 2020 Pittsburgh Spectroscopy Award from the Spectroscopy Society of Pittsburgh, the 2019 Ellis R. Lippincott Award jointly from the Optical Society of America, Society for Applied Spectroscopy and Coblentz Society, and the 2015 Craver Award from Coblentz Society. Cheng is also a Fellow of the Optical Society of America and the American Institute of Medicine and Biological Engineering.