
Philip Trackman
Professor Emeritus of Translational Dental Medicine
Research Interests
Research in Dr. Trackman’s laboratory is focused on the regulation of extracellular matrix accumulation in mineralized and non-mineralized normal tissues, and in pathologies in which extracellular matrix accumulation is affected. Studies, which utilize cell culture, animal models, and human tissues, encompass a wide range of experimental approaches derived from the disciplines of biochemistry, enzymology, cell biology, and quantitative biology.
The laboratory is investigating the mechanism by which lysyl oxidase like-2 (LOXL2) contributes to oral cancer development and metastasis. Interactions between tumor secreted LOXL2 and surrounding fibroblasts in cancer development are of primary interest at this time. The laboratory has contributed to the development of mouse models of oral cancer that are now additionally used by other researchers at the School of Dental Medicine and the School of Medicine.
The mechanism by which lysyl oxidase (LOX) acts as a tumor suppressor is under investigation. Dr. Trackman’s laboratory has made the novel discovery that the tumor suppressor function of lysyl oxidase resides in the propeptide (LOX-PP) region of a proenzyme precursor. This propeptide is released from the proenzyme by extracellular proteolytic processing, and the released propeptide inhibits growth of tumor cells and tumor formation after tumor cell uptake of LOX-PP. A focus of the laboratory is to identify mechanisms by which the lysyl oxidase propeptide can suppress tumor formation or tumor growth and tumor metastasis.
Diabetic osteopenia leads to poor mobility and morbidity in diabetics, and compromises the effectiveness of a variety of dental treatments. A focus of the laboratory is to investigate the importance and mechanism of diabetes down-regulation of lysyl oxidase as a major contributing factor to diabetic osteopenia. The current hypothesis is that the response of osteoblasts to gastric hormones known as incretins is compromised in diabetes, leading to low lysyl oxidase production, poor bone collagen cross-linking, and weak bones. In addition, the basic bone phenotypes of lysyl oxidase family deficient mice is under investigation.
Publications
Min C, Kirsch KH, Zhao Y, Jeay S, Palamakumbura AH, Trackman PC, Sonenshein GE. The tumor suppressor activity of the lysyl oxidase propeptide reverses the invasive phenotype of Her-2/neu-driven breast cancer. Cancer Res. 2007 Feb 1;67(3):1105-12
Kantarci A, Black SA, Xydas CE, Murawel P, Uchida Y, Yucekal-Tuncer B, Atilla G, Emingil G, Uzel MI, Lee A, Firatli E, Sheff M, Hasturk H, Van Dyke TE, Trackman PC. Epithelial and connective tissue cell CTGF/CCN2 expression in gingival fibrosis. J Pathol. 2006 Sep; 210(1):59-66.
Heng ED, Huang Y, Black SA, Trackman PC. CCN2, connective tissue growth factor, stimulates collagen deposition by gingival fibroblasts via module 3 and alpha6- and beta1 integrins.J Cell Biochem. 2006 May 15;98(2):409-20.
Palamakumbura AH, Trackman PC. A fluorometric assay for detection of lysyl oxidase enzyme activity in biological samples. Anal Biochem. 2002 Jan 15;300(2):245-51.
Uzel MI, Kantarci A, Hong H-H, Uygur C, Sheff MC, Firatli E, Trackman PC. Connective tissue growth factor in drug-induced gingival overgrowth. J. Periodontol. 2001.
Hong H-H, Uzel MI, Duan C, Sheff MC, and Trackman PC. Regulation of lysyl oxidase, collagen, and connective tissue growth factor by TGF-b1 and detection in human gingiva. Laboratory Investigation 79:1655-1667, 1999.
Feres-Filho EJ, Choi YJ, Han X, Takala TES, and Trackman PC. Pre- and post-translational regulation of lysyl oxidase by transforming growth factor-b1 in osteoblastic MC3T3-E1 cells. J. Biol. Chem. 270:30797-30803, 1995.
Patents
| Publication Number: | WO/2005/094424 | International Application No.: | PCT/US2005/000631 |
| Publication Date: | 13.10.2005 | International Filing Date: | 06.01.2005 |
Recent Grants
Growth Factors and Gingival Fibrosis, 8//2007-6/2012 NIH-R01
Inhibited Intramembranous Bone Healing in Diabetes, 6/2003-5/2008 NIH-R01
Lysyl Oxidase Inhibition of RAS-Mediated Transformation, 4/2004-3/2009, NIH-R01 Co-PI
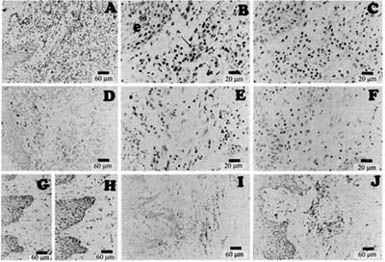
Immunohistochemistry of hyperplastic human gingival tissues from a patient undergoing treatment with Dilation (AtoH), and tissue from a patient undergoing therapy with nifedipine (I and J). Sections stained with TGF-b1 antibody are shown in A, B, and I. A and B are photos of the same section, but different magnifications. The section shown in C is stained with nonimmune IgG and is a serial section of that shown in A and B. Sections stained with CTGF antibody are shown in D, E, and J, D and E show different magnifications of the same section. The section shown in F is stained with preimmune IgG and is a serial section of that shown in D and E. The section stained with lysyl oxidase antibody is shown in G, and the serial section (H) is stained with nonimmune IgG. Hong, et al. 79 1655-1666, 1999.
- Departments
- Translational Dental Medicine
- Affiliations
- Faculty